-
Faculty of Arts and HumanitiesDean's Office, Faculty of Arts and HumanitiesJakobi 2, r 116-121 51005 Tartu linn, Tartu linn, Tartumaa EST0Institute of History and ArchaeologyJakobi 2 51005 Tartu linn, Tartu linn, Tartumaa EST0Institute of Estonian and General LinguisticsJakobi 2, IV korrus 51005 Tartu linn, Tartu linn, Tartumaa EST0Institute of Philosophy and SemioticsJakobi 2, III korrus, ruumid 302-337 51005 Tartu linn, Tartu linn, Tartumaa EST0Institute of Cultural ResearchÜlikooli 16 51003 Tartu linn, Tartu linn, Tartumaa EST0Institute of Foreign Languages and CulturesLossi 3 51003 Tartu linn, Tartu linn, Tartumaa EST0School of Theology and Religious StudiesÜlikooli 18 50090 Tartu linn, Tartu linn, Tartumaa EST0Viljandi Culture AcademyPosti 1 71004 Viljandi linn, Viljandimaa EST0Professors emeriti, Faculty of Arts and Humanities0Associate Professors emeriti, Faculty of Arts and Humanities0Faculty of Social SciencesDean's Office, Faculty of Social SciencesLossi 36 51003 Tartu linn, Tartu linn, Tartumaa EST0Institute of EducationJakobi 5 51005 Tartu linn, Tartu linn, Tartumaa EST0Johan Skytte Institute of Political StudiesLossi 36, ruum 301 51003 Tartu linn, Tartu linn, Tartumaa EST0School of Economics and Business AdministrationNarva mnt 18 51009 Tartu linn, Tartu linn, Tartumaa EST0Institute of PsychologyNäituse 2 50409 Tartu linn, Tartu linn, Tartumaa EST0School of LawNäituse 20 - 324 50409 Tartu linn, Tartu linn, Tartumaa EST0Institute of Social StudiesLossi 36 51003 Tartu linn, Tartu linn, Tartumaa EST0Narva CollegeRaekoja plats 2 20307 Narva linn, Ida-Virumaa EST0Pärnu CollegeRingi 35 80012 Pärnu linn, Pärnu linn, Pärnumaa EST0Professors emeriti, Faculty of Social Sciences0Associate Professors emeriti, Faculty of Social Sciences0Faculty of MedicineDean's Office, Faculty of MedicineRavila 19 50411 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Biomedicine and Translational MedicineBiomeedikum, Ravila 19 50411 Tartu linn, Tartu linn, Tartumaa ESTInstitute of PharmacyNooruse 1 50411 Tartu linn, Tartu linn, Tartumaa ESTInstitute of DentistryL. Puusepa 1a 50406 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Clinical MedicineL. Puusepa 8 50406 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Family Medicine and Public HealthRavila 19 50411 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Sport Sciences and PhysiotherapyUjula 4 51008 Tartu linn, Tartu linn, Tartumaa ESTProfessors emeriti, Faculty of Medicine0Associate Professors emeriti, Faculty of Medicine0Faculty of Science and TechnologyDean's Office, Faculty of Science and TechnologyVanemuise 46 - 208 51003 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Computer ScienceNarva mnt 18 51009 Tartu linn, Tartu linn, Tartumaa ESTInstitute of GenomicsRiia 23b/2 51010 Tartu linn, Tartu linn, Tartumaa ESTEstonian Marine Institute0Institute of PhysicsInstitute of ChemistryRavila 14a 50411 Tartu linn, Tartu linn, Tartumaa EST0Institute of Mathematics and StatisticsNarva mnt 18 51009 Tartu linn, Tartu linn, Tartumaa EST0Institute of Molecular and Cell BiologyRiia 23, 23b - 134 51010 Tartu linn, Tartu linn, Tartumaa ESTTartu ObservatoryObservatooriumi 1 61602 Tõravere alevik, Nõo vald, Tartumaa EST0Institute of TechnologyNooruse 1 50411 Tartu linn, Tartu linn, Tartumaa ESTInstitute of Ecology and Earth SciencesJ. Liivi tn 2 50409 Tartu linn, Tartu linn, Tartumaa ESTProfessors emeriti, Faculty of Science and Technology0Associate Professors emeriti, Faculty of Science and Technology0Institute of BioengineeringArea of Academic SecretaryHuman Resources OfficeUppsala 6, Lossi 36 51003 Tartu linn, Tartu linn, Tartumaa EST0Area of Head of FinanceFinance Office0Area of Director of AdministrationInformation Technology Office0Administrative OfficeÜlikooli 18A (III korrus) 51005 Tartu linn, Tartu linn, Tartumaa EST0Estates Office0Marketing and Communication OfficeÜlikooli 18, ruumid 102, 104, 209, 210 50090 Tartu linn, Tartu linn, Tartumaa EST0Area of Vice Rector for DevelopmentCentre for Entrepreneurship and InnovationNarva mnt 18 51009 Tartu linn, Tartu linn, Tartumaa EST0University of Tartu Natural History Museum and Botanical GardenVanemuise 46 51003 Tartu linn, Tartu linn, Tartumaa EST0International Cooperation and Protocol Office0University of Tartu MuseumLossi 25 51003 Tartu linn, Tartu linn, Tartumaa EST0Area of RectorRector's Strategy OfficeInternal Audit OfficeArea of Vice Rector for Academic AffairsOffice of Academic Affairs0University of Tartu Youth AcademyUppsala 10 51003 Tartu linn, Tartu linn, Tartumaa EST0Student Union OfficeÜlikooli 18b 51005 Tartu linn, Tartu linn, Tartumaa EST0Centre for Learning and TeachingArea of Vice Rector for ResearchUniversity of Tartu LibraryW. Struve 1 50091 Tartu linn, Tartu linn, Tartumaa EST0
Chair of Colloid and Environmental Chemistry
Main research areas
1) Studies of nanostructured and chemically modified materials (Kaido Tammeveski, kaido.tammeveski@ut.ee)
- development of new cathode materials for low-temperature fuel cells
- oxygen reduction on nitrogen-doped carbon nanomaterials and non-precious metal catalysts
- electrochemical reduction of oxygen on nanostructured metal electrodes
- electrocatalytic properties of metal nanoparticles attached to carbon nanotubes and graphene
- electrocatalytic properties of chemically modified electrodes towards oxygen reduction
- influence of organic films on the electrochemical properties of various materials (C, Au, Ni)
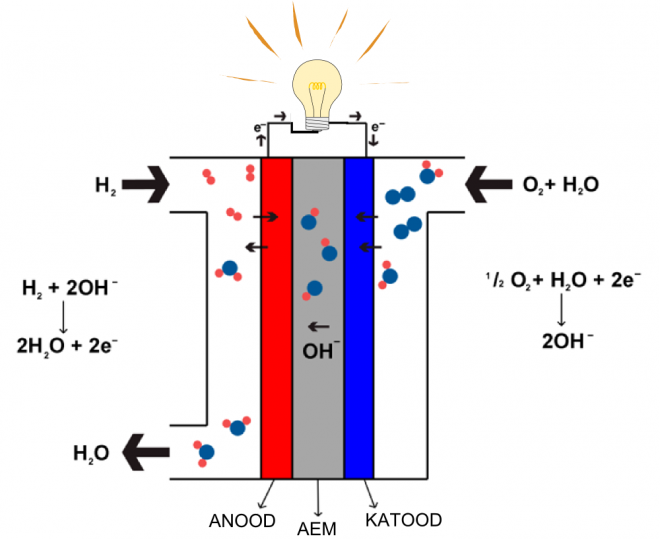
The study of electrocatalysis of the oxygen reduction reaction is related to the development of low-temperature fuel cells (see Scheme). The electrochemical reduction of oxygen occurs on the fuel cell cathode and platinum and its alloys are predominantly used as electrocatalysts for this process. The high price of Pt and its limited supply are the main factors that hinder the wider commercialization of fuel cells. For this reason we have carried out research to replace costly platinum with cheaper alternative catalysts. The basic requirements for novel and less expensive catalysts materials are high electrocatalytic activity towards oxygen reduction and good durability in fuel cell conditions. We have paid a great deal of attention to the determination of kinetic parameters for oxygen reduction on these non-precious metal catalysts.
Fuel cells, which operate at lower temperature than 100 ºC, are considered as attractive energy conversion devices in several fields, including automotive applications. Fuel cell technology is environmentally friendly, because the only product of fuel cell processes is water.
Group members:
Dr. Kaido Tammeveski, Associate Professor
Dr. Ave Sarapuu, Senior Research Associate
Dr. Nadežda Kongi, Researcher
Dr. Ivar Kruusenberg, Researcher
Dr. Elo Kibena, Researcher
In 2010-2015 four PhD thesis have been defended (Nadežda Kongi, Marko Kullapere, Ivar Kruusenberg and Elo Kibena). We have published 50 papers in peer-reviewed Journals during this period.
2) Environmental chemistry and technology (Toomas Tenno, toomas.tenno@ut.ee)
- environmental impact of oil shale semi-coke deposition and physico-chemical processes in semi-coke
- production of biogas by anaerobic treatment of biological waste
- mass transfer processes through the air-water interface
- bioremediation of soils polluted with oil products
- optimization of the composting technologies
3) Research and development of biosensors (Toonika Rinken, toonika.rinken@ut.ee)
- biosensor construction and modelling
- study of biosensor arrays
- development and application of biosensors: for the detection of antibiotic residues in raw milk, biogenic amines etc.
- separation, purification and immobilization of enzymes and other bioactive compounds for application in biosensors
- development of biosensors based on microorganisms and their application for the determination of biochemical oxygen demand (BOD)
Courses
Lectures, seminars and laboratory works in environmental chemistry, analysis and modelling, waste management, colloid- and surface chemistry, etc.


